Magnesium extraction from chromium waste streams
Introduction
Magnesium has a wide range of uses in industry from die casting and alloying to medicine.1,2 Magnesium has low density, and is seen as an important metal for applications and manufacturing where weight reduction is important. Since the beginning of this millennium, magnesium use has grown considerably mainly fuelled by China.
Magnesium is found in minerals such as dolomite, magnesite, brucite, serpentinite, and is also found in seawater, brines and bitterns.3 Dolomite, magnesite and seawater are the main sources of commercial magnesium extraction.
Newer processes have been developed to extract magnesium and produce related chemicals, such as magnesium oxide, from serpentine mine tailings, which is an opportunity for Oman given the large amounts of these waste streams that are generated during chromium mining.
Applications
Magnesium and its compounds have been used in many applications such as die casting, alloying insulation, desulphurization of molten iron, agriculture, fertilizers, pyrotechnics, aerospace, flares, refractory materials, construction, in chemicals, pharmaceuticals and other industries.
Magnesium metal applications
Figure 28‑1 shows the main applications for magnesium metal. Die casting and aluminium alloys together make up two-thirds of where magnesium metal is used. Although magnesium is about twice as expensive as aluminium, its hot-chamber die-casting process is easier, more economical, and 40% to 50% faster than cold-chamber process required for aluminium. Another advantage of magnesium is it is a relatively low-cost option for achieving weight reduction. A comparison of weight reduction achieved by magnesium is shown in Table 28‑1. As can be seen the use of magnesium in automotive parts can achieve weight reductions greater than even aluminium.
The addition of magnesium metal in alloys increases strength and strain hardening ability. These alloys are non-heat-treatable alloys and are used for structural applications. Some of the applications include buildings, pressure vessels, train and truck bodies, chemical tankers, and armoured vehicles.
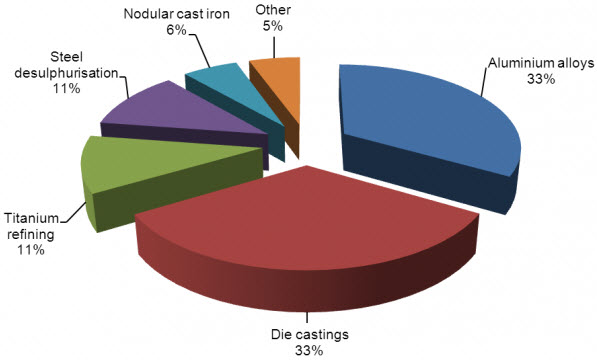
Figure 28‑1: Magnesium metal uses
Table 28‑1: Automotive part weight reduction versus low carbon steel1
|
Material |
Weight reduction vs low-carbon steel |
|
High-strength steel |
15-25% |
|
Glass-fibre composite |
25-35% |
|
Aluminium |
40-50% |
|
Magnesium |
55-60% |
|
Carbon-fibre composite |
55-60% |
Magnesium chemicals applications1
Magnesium chemicals have a variety of applications including refractories, agriculture (both as fertilizer and in feed), for magnesium metal production, water treatment, de-icing, flue gas desulphurization, as a flame retardant, pulp and paper production, pharmaceuticals and food manufacture, and in pigments and paints. Magnesium oxide is the most commercially important of the magnesium chemicals.
Market size
Magnesium metal market size
The global magnesium metal market was valued at $3.3 billion in 2020 and is predicted to reach $6.8 billion by 2027 at a CAGR of 8.8%. Increasing die casting requirements in automotive and various structural applications is projected to assist in the growth of the market.
In terms of revenue, die casting application is projected to grow the most with a CAGR of 9.0% in the metal magnesium market from 2020 to 2027. Demand from aerospace and defence applications are projected to provide lucrative opportunities for market vendors. Some of the key applications include cast gearboxes for helicopters, auxiliary casing, and transmission components.
Magnesium chemicals market size
Amongst magnesium chemicals, magnesium oxide is the most commercially important one. China dominates the market as both the largest producer and consumer of this chemical. In 2019, >50% of magnesium oxide exported from China were refractory magnesia for use in steel manufacture and cement production. The remainder was caustic-calcined magnesia, destined for environmental, construction and agricultural markets. Global use of magnesium oxide is set to increase by an average of 1.5% per year. Consumption of magnesium oxide based on geography is shown in Figure 28‑2.
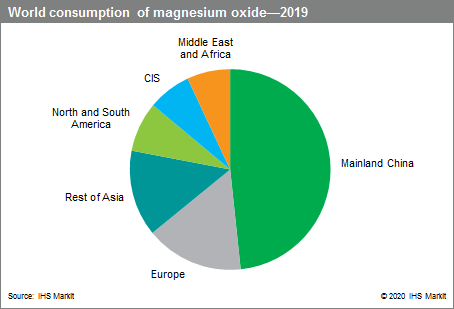
Figure 28‑2: Global consumption of magnesium oxide.
Relevance to Oman
Chromium (and other) mining in Oman produces considerable amounts of tailings. Some of this is likely to be in the form of serpentinite. Serpentinization is a form of low-temperature metamorphism of ultramafic rocks, such as dunite, harzburgite or lherzolite. These are rocks low in silica and composed mostly of olivine, pyroxene, and chromite. Serpentinization is driven largely by hydration and oxidation of olivine and pyroxene to serpentine minerals, brucite and magnetite.1 Infiltration of CO2-bearing fluids into serpentinite causes distinctive talc-carbonate alteration. Brucite rapidly converts to magnesite and serpentine minerals (other than antigorite) are converted to talc. Magnesite is a source of magnesium metal, which can be produced using well established production methods (discussed below). Magnesite can also be converted to magnesium oxide, which, as discussed above, is commercially important. The production process for magnesium oxide is also briefly discussed below.
The feasibility of extracting magnesium or producing magnesium oxide, other chemicals and other magnesium-containing products from chromium mine waste streams in Oman will need to be determined following evaluation of the tailings and determining the concentration of magnesium and other impurities present.
